By combining advanced data-driven scenario modeling, direct engagement with investigators and research teams across the Paradigm Health network, and deep clinical and regulatory expertise, we identify opportunities to reduce site and patient burden, improve study feasibility, and accelerate the path from startup to study execution.
In an effort to meet perceived regulatory expectations, protocols have become increasingly complex, often introducing eligibility criteria, data collection requirements, and study procedures that fail to reflect standard clinical workflows and patient realities. When protocols become misaligned with how care is actually delivered, sites are reluctant to participate, operational burden increases, and timelines slip. The result is mounting pressure to add sites or amend protocols midstream, adding cost, complexity, and strain to already resource-intensive trials and overburdened site staff.
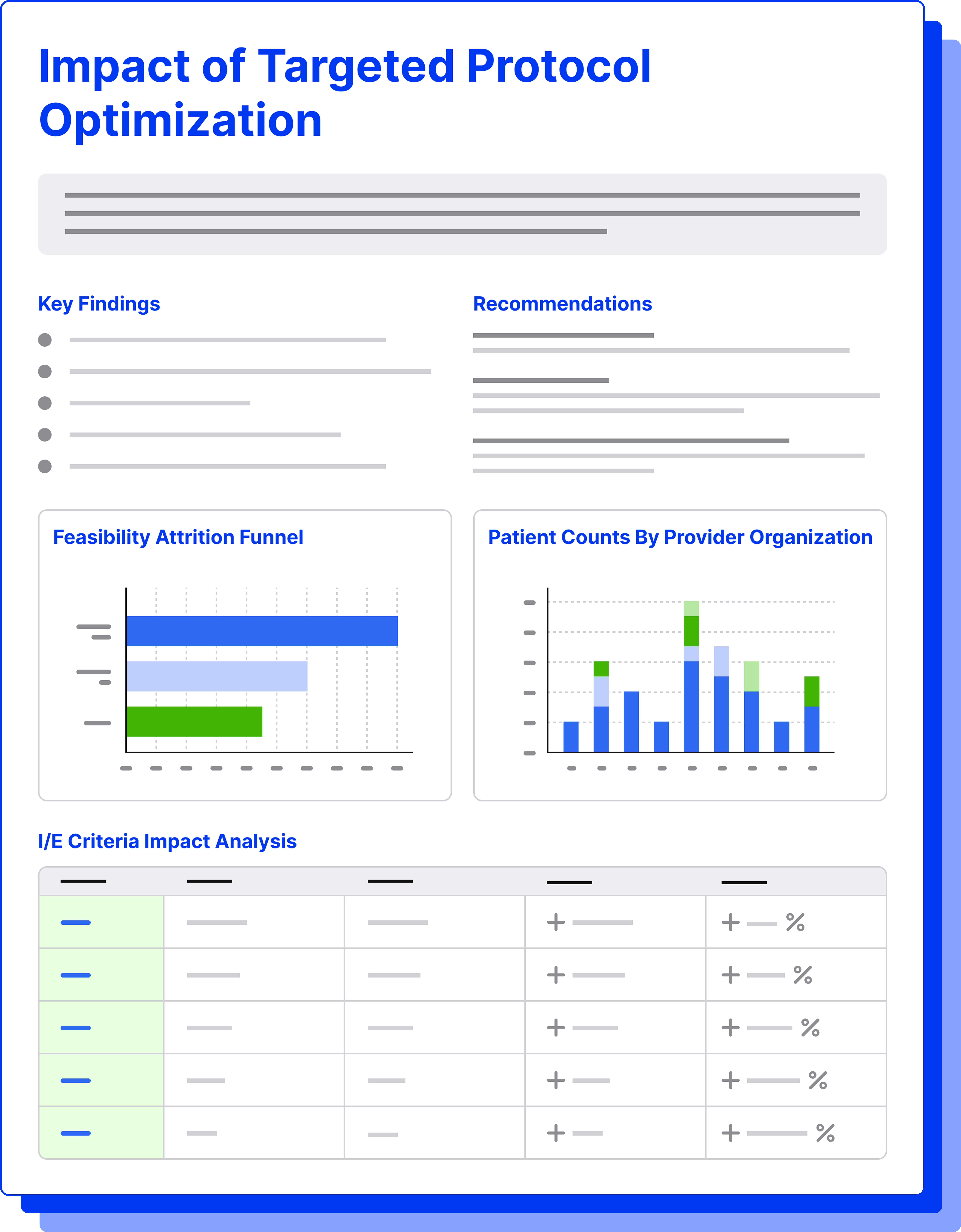
Study procedures, visit cadence, biomarker requirements, and data collection expectations that diverge from routine clinical workflows can reduce site willingness to participate and increase burden for providers and patients.
Protocol optimization, feasibility, and site selection are often treated as separate sequential activities, delaying startup and limiting opportunities to refine protocols based on direct site feedback before activation. The result can be operational friction during study conduct, increased burden on sites and patients, and a greater risk of protocol deviations.
Paradigm Health helps sponsors optimize clinical trial protocols for operational feasibility, startup readiness, and efficient study execution. Through quantitative modeling, clinical and regulatory review, and direct engagement with investigators and research teams, we identify operational and enrollment risks before study startup and deliver structured recommendations to improve protocol fit across diverse care settings.
Unlike traditional approaches where protocol optimization, feasibility, site selection, and activation occur as separate sequential activities, Paradigm Health incorporates operational and site-level feedback earlier in the process—helping sponsors improve site willingness to participate, reduce downstream friction, and accelerate the transition from startup to study execution.
Paradigm Health combines data-driven scenario modeling, clinical review, and direct engagement with investigators and research teams to identify operational, enrollment, and execution risks before study startup. Findings are delivered through structured feasibility and optimization reports designed to support protocol refinement, site readiness, and downstream study execution.
Model eligibility criteria to quantify impact on recruitable patient volume, demographic representation, and site-level distribution while refining protocol language to improve interpretability for participating sites.
Engage investigators, coordinators, and research teams across the Paradigm Health network to identify feasibility risks, operational barriers, and opportunities to improve site participation before activation.
Assess visit cadence, study procedures, and operational workflows against standard clinical practice to reduce site and patient burden while improving execution readiness.
Evaluate protocol-required data collection, assessments, and CRF structure to streamline study conduct, reduce operational burden, and support efficient downstream monitoring and EHR-integrated data capture.
Evaluate biomarker requirements and testing workflows to ensure efficient patient identification, reduce operational friction, and improve enrollment readiness across sites.
Pressure-test protocol assumptions against current clinical practice, regulatory guidance, and operational realities to reduce amendment risk and strengthen execution feasibility.
By aligning protocol strategy, operational workflows, data collection requirements, and site feedback with clinical and regulatory expectations, Paradigm Health helps sponsors reduce friction before study startup, improve site participation, and execute with fewer downstream operational challenges.
Direct engagement with investigators and research teams during protocol optimization helps identify operational risks earlier, improve site willingness to participate, and pull key elements of feasibility and site selection forward, meaningfully accelerating the path from design to first patient in.
Eligibility, biomarker, and workflow optimization expand the addressable patient pool, improve operational fit across sites, and support broader, more representative enrollment across diverse care settings.
Early identification of operational and clinical friction points helps reduce unnecessary procedures, streamline data collection requirements, minimize downstream amendments, and improve execution efficiency for sites and sponsors.
Whether you're designing a new study or preparing for activation, early protocol decisions shape enrollment performance, site adoption, and amendment risk. Connect with Paradigm Health to assess feasibility, strengthen execution readiness, and move forward with greater confidence.
Let’s build a protocol positioned for performance.